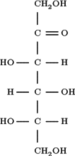
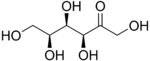
Sorbose
| |||
| Names | |||
|---|---|---|---|
| IUPAC name L-xylo-Hex-2-ulose | |||
| Systematic IUPAC name (3S,4R,5S)-1,3,4,5,6-Pentahydroxyhexan-2-one | |||
| Other names Sorbinose L-xylo-Hexulose | |||
| Identifiers | |||
CAS Number |
| ||
3D model (JSmol) |
| ||
| ChemSpider |
| ||
| ECHA InfoCard | 100.001.611 | ||
PubChem CID |
| ||
| UNII |
| ||
InChI
| |||
| |||
| Properties[1] | |||
Chemical formula | C6H12O6 | ||
| Molar mass | 180.156 g·mol−1 | ||
| Appearance | white solid | ||
| Density | 1.65 g/cm3 (15 °C) | ||
| Melting point | 165 °C (329 °F; 438 K) | ||
Solubility in water | Highly Soluble | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).  N verify (what is N verify (what is  Y Y N ?) N ?) Infobox references | |||
Chemical compound
Sorbose is a ketose belonging to the group of sugars known as monosaccharides. It has a sweetness that is equivalent to sucrose (table sugar).[1] The commercial production of vitamin C (ascorbic acid) often begins with sorbose. L-Sorbose is the configuration of the naturally occurring sugar. It can be prepared from inexpensive O-benzylglucose.
Synthesis
Under conditions employed for a Meerwein-Ponndorf-Verley reduction, the tetra-O-benzyl aldose converts to tetra-O-benzylsorbose. Hydrogenolysis removes the four benzyl groups, leaving sorbose.[2]
References
- ^ a b Merck Index, 12th Edition, 8874
- ^ Frihed, Tobias Gylling; Bols, Mikael; Pedersen, Christian Marcus (2015). "Synthesis of l-Hexoses". Chemical Reviews. 115 (9): 3615–3676. doi:10.1021/acs.chemrev.5b00104. PMID 25893557.
- v
- t
- e
Types of carbohydrates
| Dioses |
|
|---|---|
| Trioses |
|
| Tetroses |
|
| Pentoses |
|
| Hexoses | |
| Heptoses |
|
| Above 7 |
| Disaccharides | |
|---|---|
| Trisaccharides | |
| Tetrasaccharides | |
| Other oligosaccharides |
|
| Polysaccharides |
 Category
Category
 | This article about a ketone is a stub. You can help Wikipedia by expanding it. |
- v
- t
- e