Nilestriol
 | |
| Clinical data | |
|---|---|
| Trade names | Wei Ni An |
| Other names | Nylestriol; LY-49825; Ethinylestriol cyclopentyl ether; EE3CPE; 17α-Ethynylestriol 3-cyclopentyl ether |
| Routes of administration | By mouth |
| Drug class | Estrogen; Estrogen ether |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID |
|
| ChemSpider |
|
| UNII |
|
| KEGG |
|
| ChEMBL |
|
| CompTox Dashboard (EPA) |
|
| Chemical and physical data | |
| Formula | C25H32O3 |
| Molar mass | 380.528 g·mol−1 |
| 3D model (JSmol) |
|
| |
InChI
| |
Nilestriol (INNTooltip International Nonproprietary Name) (brand name Wei Ni An; developmental code name LY-49825), also known as nylestriol (USANTooltip United States Adopted Name, BANTooltip British Approved Name), is a synthetic estrogen which was patented in 1971[1] and is marketed in China.[2][3] It is the 3-cyclopentyl ether of ethinylestriol, and is also known as ethinylestriol cyclopentyl ether (EE3CPE).[4] Nilestriol is a prodrug of ethinylestriol, and is a more potent estrogen in comparison.[4] It is described as a slowly-metabolized, long-acting estrogen and derivative of estriol.[5][6] Nilestriol was assessed in combination with levonorgestrel for the potential treatment of postmenopausal osteoporosis, but this formulation ultimately was not marketed.[7]
See also
References
- ^ Official Gazette of the United States Patent and Trademark Office: Patents. U.S. Department of Commerce, Patent and Trademark Office. 1975. p. 1677.
- ^ Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 891–. ISBN 978-1-4757-2085-3.
- ^ "Nilestriol". Drugs.com.
- ^ a b McGuire W (14 December 2013). Experimental Biology. Springer Science & Business Media. pp. 161–. ISBN 978-1-4757-4673-0.
- ^ Schoenberg DR (1977). Biochemical Properties of the Cytoplasmic Estrogen Receptors from Immature Rat and Mature Rabbit Uteri (Ph.D. thesis). University of Wisconsin. p. A-17.
- ^ "Section 10: Obstetrics and gynecology". Excerpta Medica. 1978.
- ^ Aronson JK (21 February 2009). Meyler's Side Effects of Endocrine and Metabolic Drugs. Elsevier. pp. 173–. ISBN 978-0-08-093292-7.
- v
- t
- e
- #WHO-EM
- ‡Withdrawn from market
- Clinical trials:
- †Phase III
- §Never to phase III
- See also
- Estrogen receptor modulators
- Androgens and antiandrogens
- Progestogens and antiprogestogens
- List of estrogens
 | This drug article relating to the genito-urinary system is a stub. You can help Wikipedia by expanding it. |
- v
- t
- e
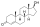 | This article about a steroid is a stub. You can help Wikipedia by expanding it. |
- v
- t
- e












